


About the Book
A book of essays and short stories in three parts and ten chapters about the people and the achievements of the great science of the structure and development of the Universe from antiquity to the present day, composed and written on the basis of many sources by the author.
About the Author
A. Tomilin is a lecturer at an institute. Engaging Essays on Cosmology is not the first book by this author.
Young readers have repeatedly encountered his name in the pages of journals and popular
science collections. Several works have come from his pen, published by the “Children’s
Literature” publishing house, including the booklet For What – Nothing, written in
collaboration with N.V. Terebinskaya, the book Project Alpha K–2, as well as the book Engaging Essays on Astronomy, issued by “Molodaya Gvardiya.”
This English translation of the book in English by Damitr Mazanav, has an extended epilogue containing exciting new discoveries the theories from 1970s to 2026 that have fundamentally changed our views about Universe.
Translated from the Russian and typeset in LaTeX by Damitr Mazanav using Quattrocento
Illustrations: G. Kovanov and V. Kovynev
This English translation released on the Web by The Mir Titles Project in 2026
under Creative Commons Share Alike 4.0 License.
You can get the book here and here
Translator’s Note
This is the fourth book I am translating the Eureka series. The author A.N.~Tomilin has written several popular science books. We have earlier seen his book on cosmogony, titled Fascinating Cosmogony. This book is on a related theme of Cosmology. I was first introduced to ideas of cosmology and general relativity during my masters. Since then, the idea that we can infact study and make sense of the origins and structure of the universe at the largest scale have fascinated me. Translating this book in many ways was refreshing some of the ideas of cosmology and general relativity that I had learned. The quote by Corliss is indicative of how with limited data, we here on Earth, an insignificant part of the universe can make sense of it at all, constructing theories that will make sense of what we observe.
Since the book was written in 1971, several speculative hypotheses presented in the book have not been supported by data. To indicate this, at some places I have added sidenotes with my initials “DM”. I have also added an \textbf{Epilogue: Cosmology as of 2026} which summarises some of the major landmarks in the themes discussed since the book was written in 1970s. Another change that I have added is to give the chapters descriptive titles. Originally, the chapters were just numbered, but I thought giving descriptive titles would be a better choice.

The reader will occasionally encounter brief, aphoristic remarks attributed to Kozma Prutkov in the last chapter. These are not incidental embellishments, but deliberate inclusions. Kozma Prutkov was not a single individual, but a fictional author created in nineteenth-century Russia by a group of writers, including Aleksey Tolstoy and the Zhemchuzhnikov brothers. Presented as a pompous yet self-assured thinker, Prutkov’s writings consist of concise maxims, paradoxes, and satirical observations on knowledge, reason, and human pretension.
At first glance, such a figure may seem out of place in a discussion of cosmology. Yet the inclusion is not without purpose. Scientific inquiry, particularly in a field as vast and abstract as cosmology, carries with it a natural tendency towards overconfidence in models, measurements, and interpretations. Prutkov’s aphorisms serve as a quiet counterpoint to this tendency. They remind us — often with a touch of irony — that clarity is hard-won, that appearances can mislead, and that systems of thought, however elegant, may conceal their own limitations.
There is also a more practical reason. Cosmology deals with scales and concepts far removed from everyday experience. At such distances, both literal and intellectual, language can easily become either overly technical or excessively grand. A well-placed aphorism has the virtue of restoring proportion. It compresses an idea into a form that can be held in mind, even as the surrounding discussion expands beyond immediate intuition.
The remarks of Kozma Prutkov are therefore included not as commentary on specific results, but as companions to them. They are meant to be read not as authorities, but as provocations — brief interruptions that invite the reader to pause, reflect, and occasionally question the very framework within which the narrative unfolds. Mistakes and omissions (and I am sure they will be there), if any, are my own.
Contents
Translator’s Note vii
Instead Of An Introduction xvii
I. People 1
1. The Miletus Manifesto 3
1.1. When the Earth Was Flat… 4
1.2. In the Homeland of Science .
1.3. Thales of Miletus 11
1.4. Heraclitus of Ephesus 21
1.5. Philosophy + Mathematics = ? 24
1.6. Elea: Xenophanes and Parmenides 31
1.7. Zeno of Elea and His Paradoxes 35
1.8. The Atomists 40
1.9. Summa summarum of Greek materialism 43
2. The Philosopher King of Physics
2.1. Aristotle 47
2.2. The Triumph of Aristotelianism 53
2.3. At the Decline of Classical Greco-Roman Culture 58
2.4. Zigzag of History 64
2.5. Flames of Bonfires Do Not Dispel the Night 67
2.6. The Principles of Papal Infallibility 70
2.7. The Catholic Church and Progress 75
2.8. The Middle Ages and Technology 80
3. From Cusa to Calculus 89
3.1. Nicholas of Cusa 91
3.2. Cosmological Rubicon 96
3.3. The Immortality of the Great Heretic 102
3.4. Eppur si muove!. 110
3.5. René Descartes (Cartesius) 124
3.6. Isaac Newton 132
3.7. How Useful It Is When There Are No Great Discoveries 136
3.8. Newton’s Apple 142
3.9. The Universe of Sir Isaac 148
II. Successes and Doubts 157
4. The Star-Struck Philosophers 159
4.1. The World in Lomonosov’s Hands 162
4.2. The Universe as a System — Early Speculations 166
4.3. Immanuel Kant — Natural Philosopher 170
4.4. Epilogue of the Philosopher’s Life 178
4.5. Johann Lambert 183
4.6. William Herschel 187
5. The Dark Night of the Paradox 195
5.1. Olbers’ Paradox 198
5.2. Knights of the “Heat Death” 203
5.3. Hugo von Seeliger 210
5.4. A Celestial Census Saves the Situation 213
6. The Copernicus of Curves 219
6.1. What Euclid Taught 222
6.2. The King Midas of the Land of Mathematics 229
6.3. Visiting the Flatfolk 235
6.4. Gauss’s “Magnificent Theorem” 241
6.5. The Copernicus of Geometry 245
6.6. The Real Construction of the “Imaginary World”256
6.7. The Astonishing Spaces of Bernhard Riemann 261
III. Ideas 269
7. The Symphony of Space-Time 271
7.1. Can a “Decisive Experiment” Fail? 273
7.2. Overture to the Symphony of Relativity 279
7.3. In Search of the Harmony of the Universe 286
7.4. The Fourth Dimension 293
7.5. Special Theory of Relativity 302
7.6. The next step was inevitable… 305
7.7. “Look!” 314
8. The Priest, the Prize, and the Point of Origin 331
8.1. Nineteen Hundred And Seventeen, February 332
8.2. “I Only Solve The Equations” 342
8.3. Yet Another Great Discovery 354
8.4. Horizons of the Universe 365
8.5. Father Georges Solves Equations 369
8.6. Genius of George Gamow 372
8.7. “Big Bang” 377
9. A Journey Along the t-Axis 387
9.1. From “Radio Stars” to Star-like Objects 389
9.2. Quasars, or What Practice Does to Theory 395
9.3. A Journey Along the t-Axis 399
9.4. The Universe, the Year 1971 407
10. The Battle for the Soul of the Cosmos 413
10.1. When an Idea Is Not Mad Enough 415
10.2. Kabbalistics of the Twentieth Century 424
10.3. Matter + Antimatter = ? 434
10.4. The Battle of Ideas Continues 441
10.5. What Is the Universe? 446
IV. Epilogue: Cosmology as of 2026 453
11. The Dark Side of the Cosmos 455
11.1. The Mystery of the Missing Mass 457
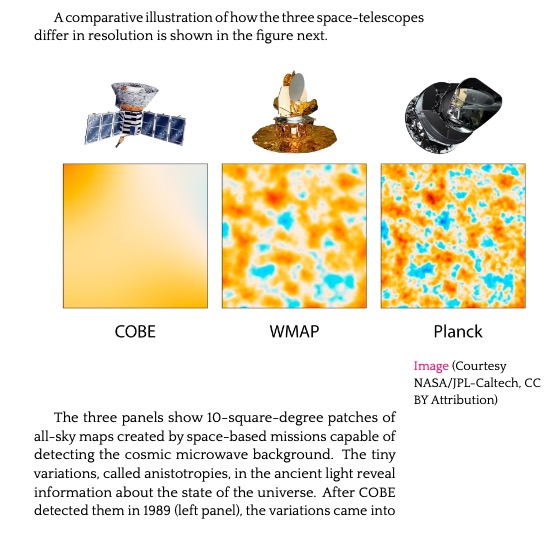
11.2. A Hi-Res Echo of the Beginning 460
11.3. WMAP and the Age of Precision 465
11.4. Planck: The Ultimate Map (So far…) 467
11.5. Seconds after the Bang 470
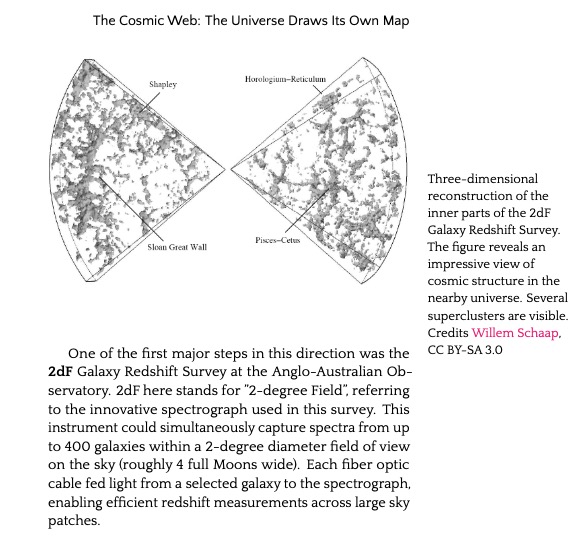
11.6. The Cosmic Web: The Universe Draws Its Own Map 474
11.7. The Sloan Digital Sky Survey 476
11.8. Dark Energy and the Runaway Universe 479
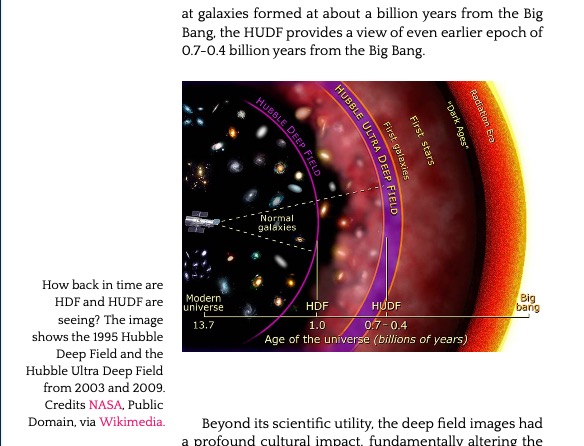
11.9. The Hubble Deep Field 483
11.10. Hearing the Black Holes 487
11.11. The Future of the “t-Axis” 490
11.12. The Hubble Tension 496
11.13. The Next Horizon: The Engines of Discovery 499
11.14. The Mathematical Horizon 503
11.15. Concluding Thoughts 504


















